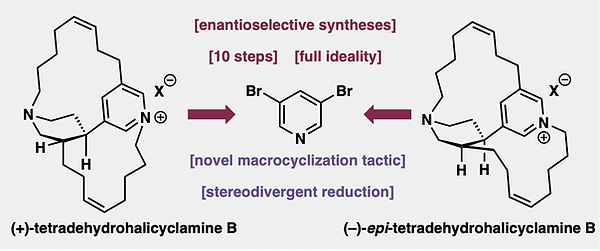
25. Firestone, Z. D.; Grigolo, T. A.; Pernichelle, F. G.; Smith, J. M.; "Enantioselective Total Synthesis of Two Aromatized Halicyclamines" J. Am. Chem. Soc., 2026, 148, 6814–6819. Chem Arxiv preprint.
24. London, H. C.; Chen, J.; Fosu, E. A.; Firestone, Z. D.; Lambert, E. C.; Smith, J. M.; Jakubikova, E.; Hanson, K. "Modulating Excited State Lifetimes in Cu(I) Complexes: The Role of Surface Binding Motifs" ACS Appl. Energy Mater. 2025, ASAP.
23. Knight, B. J.; Grigolo, T. A.; Tolchin, Z. A.; Smith, J. M. "Azine Dearomatization in Natural Product Total Synthesis " Chem. Eur. J. 2025, 31, e202402413.
22. Smith, J. M. "Heterocyclic Surgery for Isotopic Labeling" Synlett 2025, 36, 601–606 (invited contribution).
21. Dukes, D. M.; Atanassov, V. K.; Smith, J. M. "Enantioselective Total Synthesis of (+)-Cylindricine B" Chem. Sci. 2024, 15, 16554–16558.
20. Tolchin, Z. A.; Smith, J. M. " NRORC: An Azine Labeling Protocol" J. Am. Chem. Soc. 2024, 146, 2939–2943.
Highlighted by Chemistry Views
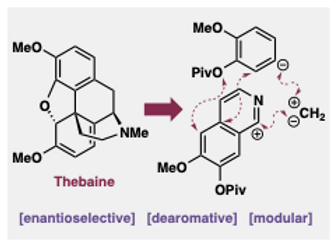
19. Tolchin, Z. A.; Dukes, D. M.; Gharbaoui, L. M.; Smith, J. M. "Dearomative Access to (–)-Thebaine and Derivatives " Org. Lett. 2023, 25, 8424–8428.
Highlighted by OPRD
Featured on: Synfacts
18. Knight, B. J.; Harbit, R. C.; Smith, J. M. "A Six-Step Synthesis of (±)-Lysergic Acid" J. Org. Chem. 2023, 88, 2158–2165.
To view a preprint, see: Chem Rxiv.
Featured on: Synfacts
Highlighted by Total Synthesis on YouTube
17. Piwko, A. T.; Miller, B. G.; Smith, J. M. "Revisiting the Manzamine Biosynthetic Hypothesis" Nat. Prod. Rep. 2023, 40, 964–971.
16. Grigolo, T. A.; Smith, J. M. "Regiodivergent Asymmetric Pyridinium Additions: Mechanistic Insight and Synthetic Applications" Chem. Eur. J. 2022, 28, e202202813.
Featured on: Synfacts
15. Grigolo, T. A.; Subhit, A. R.; Smith, J. M. "Regioselective Asymmetric Alkynylation of N–Alkyl Pyridiniums" Org. Lett. 2021, 23, 6703–6708.
Featured on Organic Chemistry Portal.
To view a preprint, see:ChemRxiv 2021.
14. Knight, B. J.; Tolchin, Z. A.; Smith, J. M. "A Predictive Model for Additions to N-Alkyl Pyridiniums" Chem. Commun. 2021, 57, 2693–2696.
To view a preprint see: ChemRxiv 2020.
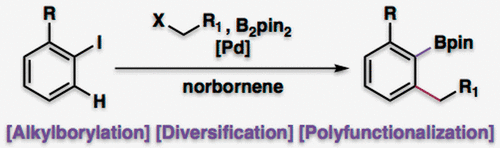
13. Sui, X.; Grigolo, T. A.; O'Connor, C. J.; Smith, J. M. "Ortho/Ipso Alkylborylation of Aryl Iodides" Org. Lett. 2019, 21, 9251–9255.
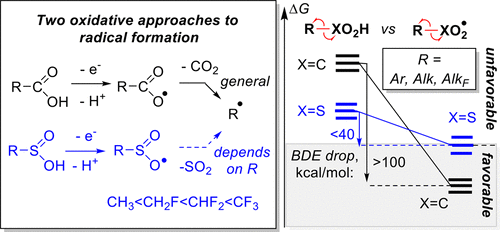
12. Gomes, G. P.; Wimmer, A.; Smith, J. M.; König, B.; Alabugin, I. V. "CO2 or SO2 : Should It Stay or Should it Go?" J. Org. Chem. 2019, 84, 6232–6243.